Turning Defects Into Profits:
The Quality Excellence Program (QEP) drives defect rates down to better meet customer requirements, while generating savings from reducing non-performance cost. Embarking on Quality Excellence (QI) is critically important when failure rate is excessive or warranty obligations hurt profitability. The program helps quality managers, business leaders, and functional specialists to systematically improve Process Capability and consistently meet customer requirements at lowest quality cost.
Quality Excellence Program
Why Is Quality Important?
Quality is critical to satisfy customers and keep costs under control. Superior products and services command higher prices and significantly contribute to long-term revenue and profitability, making quality is a key differentiator in a crowded market. Continuous quality improvement is therefore a prerequisite for long-term success.
What is Quality Excellence?
Quality implies a certain level of success in meeting customer needs, creating products and services that meet specifications and expectations, and comply with applicable standards. Excellence refers to an internal drive to become the best. Quality Excellence (QE) therefore, is the drive to become a leader in quality.
What Are the Challenges?
The two Quality Challenges faced by any manufacturer and service provider are (a) Reducing variability when customer requirements are not met and (b) Reducing quality cost when customer requirements are consistently being met, and process capability is established. Solving these two quality challenges is an investment in the future, and a top priority when failures erode profitability and undermine long-term sustainability.
What Is Quality Actually?
Quality is a degree of excellence, the fitness for intended use, the ability to fulfill customer needs (→Quality Philosophy). Non-quality therefore is any deviation from the specification, expectations, or standard, such as a rejected proposal, missed goal, overdue decision, late delivery, lost order, healthcare failure, or returned package. Defects hurt profitability in the short-term and erode competitiveness long-term. Quality champions consider defects as opportunities, improving bottom-line profitably by eliminating poor quality.
What Is a Quality Excellence Program? How Does It Work?
To be effective, a quality program must address the causes for variability, defects, and the quality costs. Our Quality Excellence Program (QEP) helps client organizations improve performance, move towards zero-defect capability by addressing excessive complexity, process instability, ineffective control, and the eight causes of human failure. It establishes the critical-to-quality (CTQ) parameters that must be controlled to ensure consistent outcomes within specified tolerances, while reducing the cost of poor quality (COPQ) that elevates the bottom line.
How Long Does the Program Take?
By using a series of analysis and trials, straight-forward problems can be addressed within a single quality improvement sprint; a week to optimize one critical-to-quality parameter and several weeks to address interaction effects. Broad and deep-rooted problems require several weeks to identify and eliminate root-causes at the physical, human, and latent level →Problem Solving. A 90-day project allows addressing a complete cross-functional failure tree at the corporate level. As a rule of thumb, we schedule one month per function, entity, or major stakeholder involved.
Who Must Be Involved? Who Is Leading?
Functional managers, process experts, and experienced operators are key members of the quality improvement team. A project sponsor and project controller must be assigned to drive the program internally, while Leanmap provides external support. Acting as an analyst, problem solver, or interim quality leader (→Engagement Modal), we provide the critical knowledge and fill resource gaps to bring quality in line with expectations.
How Much Does it Cost?
Defects hurt profitability in the short term and erode competitiveness in the long term. Consequently, every root-cause eliminated and every error-trap implemented reduces quality costs. Our Quality Excellence Program (QEP) is not only self-funding but contributes to the bottom line by generating savings from fewer defects, freeing up people, capacity, and capital (e.g. released buffer and safety stock). For the support we provide, every dollar you spend in consulting fees, we identify 10x to 30x in quality improvement potentials. Typical ROI of our quality-focused improvement projects is 11x (→ 6D Process).
What Are the Benefits?
Quality improvement effort pays off. Our Quality Excellence Program (QEP) creates better outcomes for all stakeholders: better products for customers, better care for patients, stable processes for employees, and healthy returns for investors. Training your staff in process control and systematic problem solving ensures the critical skills are in place to continuously improve quality, cutting defect rates in half each year, until the desired level of Quality Excellence is achieved.
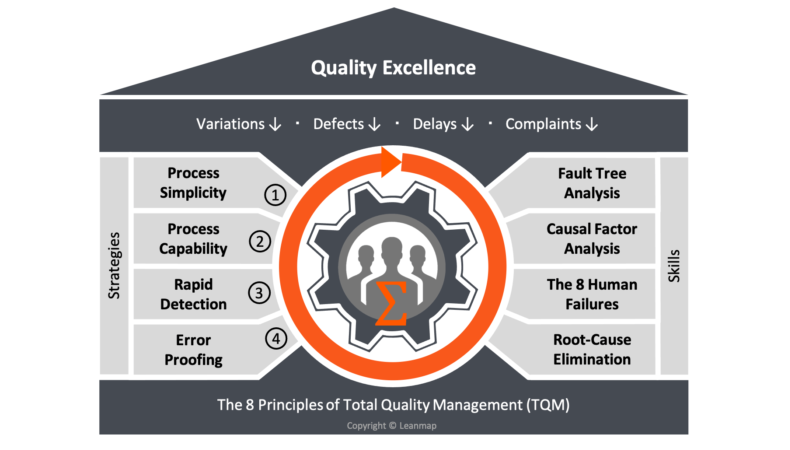
The 4 Quality Improvement Strategies Reduce Defect Rate by 1000x
The path to Quality Excellence requires implementing four distinct strategies, each reducing defect rates by 10x, or 1000x overall. Applying these strategies dramatically improves the quality of manufacturing and service processes, including product development, customer service, transportation, utilities, and healthcare.
1. Process Simplicity
➤ Defects increase with the difficulty and duration of a task. Process complexity is therefore a cause of both, variability and mistakes, that result in defects.
➤ By changing the process so that fewer or simpler steps are needed, we reduce the probability of omitting a critical step or uncontrolled variation in a procedure.
➤ Simplification reduces defects at their source. It must occur first because it is extremely difficult to change a process after it has been institutionalized. It is the main reason why simplification is addressed ahead of variability, speed, and mistakes to bring defect rate to 10% or less.
2. Process Capability
➤ Defects increase with variability from instable processes and uncontrolled interactions with the operating environment, such as the inability to hold tolerances.
➤ Variability is addressed using statistical methods (SQC, SPC) to make processes more stable. It requires an understanding of the science behind processes and also the analysis of variance.
➤ Establishing process capability by controlling the characteristics of the product or service, while eliminating the causes of excessive variations brings defect rate down from 10% to 1%.
3. Rapid Detection
➤ Defects increase with batch size and problem-detection time. The longer it takes to catch a problem, the more often a defect can be reproduced.
➤ Problem detection time is minimal when moving from batches to sequential processing (FIFO) using cells and flow lines. It provides instant feedback on infrequently occurring discrete events like tool breakage or mislabeled parts.
➤ Separating unprocessed from completed work prevents reproducing defects, while rapid detection brings defect rate down from 1% to 0.1%.
4. Error-Proofing
➤ Defects are also caused by human mistakes and errors, such as omitting steps, doing them incorrectly, or processing them in the wrong order.
➤ Mistakes cannot be controlled by statistical methods that focus on variability. We therefore need to implement error-traps, go/no-go fixtures, and inline validation checks.
➤ Because mistakes are inevitable and their consequences are often severe, they must be prevented. Protecting the process from human mistakes, using error-traps and validation checks brings defect rate further down from 0.1% to 0.01%.
Total Quality Principles Are the Foundation of any Quality Improvement Program
What is Total Quality Management?
➤ Total implies that all employees of an organization are involved to improve all processes, from identified need to its fulfillment.
➤ Quality is fitness for intended use, covering all features and characteristics of a product or service to satisfy customer needs.
➤ Management refers to a focused effort, providing funding, training, staffing, direction to manage outcomes and achieve goals.
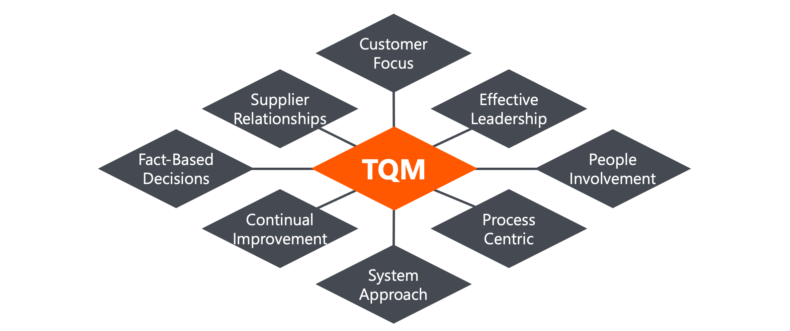
The 8 Principles of TQM
To promote a total quality culture in the client-organizations we work with, we align our programs with the eight Total quality Management principles that have been defined by the International Organization for Standardization (ISO), the Technical Committee 176 (TC 176), responsible for Quality Management and quality assurance. Those principles are:
Principle 1: Customer Focus
Organizations depend on their customers, must understand and meet their customers current and future needs.
➤ Understand their needs
➤ Meet their requirements
➤ Strive to exceed their expectations
Principle 2: Effective Leadership
Leaders establish unity of purpose and direction, while creating an environment in which people can achieve goals.
➤ Establish direction
➤ Provide training
➤ Ensure funding and staffing
Principle 3: People Involvement
Get all people engaged and committed, so they can fully contribute, ensuring that their abilities are used for the organization’s benefit.
➤ Involve everyone
➤ Empower people
➤ Create a conducive work environment
Principle 4: Process Centric
The desired result is achieved more efficiently when related resources and activities are managed as a process.
➤ Establish process capability
➤ Systematically reduce variations
➤ Monitor performance to detect abnormalities
Principle 5: System Approach
Identifying, understanding, and managing interrelated processes improves the organization’s effectiveness and efficiency.
➤ Standardize processes
➤ Establish rules and limits
➤ Address deviations
Principle 6: Continual Improvement
Never-ending improvement must be a permanent objective of an organization to sustain success in the long term.
➤ Challenge the status quo
➤ Evolve best-known practices
➤ Raise target faster than competitors
Principle 7: Fact-Based Decisions
Effective decisions and actions are based on the analysis of data and information.
➤ Strategy based on opportunities and tradeoffs
➤ Tactics based on projected return on investment
➤ Operational decisions based on performance data
Principle 8: Supplier Relationships
An organization and its suppliers are independent, and a mutually beneficial relationship enhances the ability to create value.
➤ Create partnerships
➤ Align strategies
➤ Connect value streams
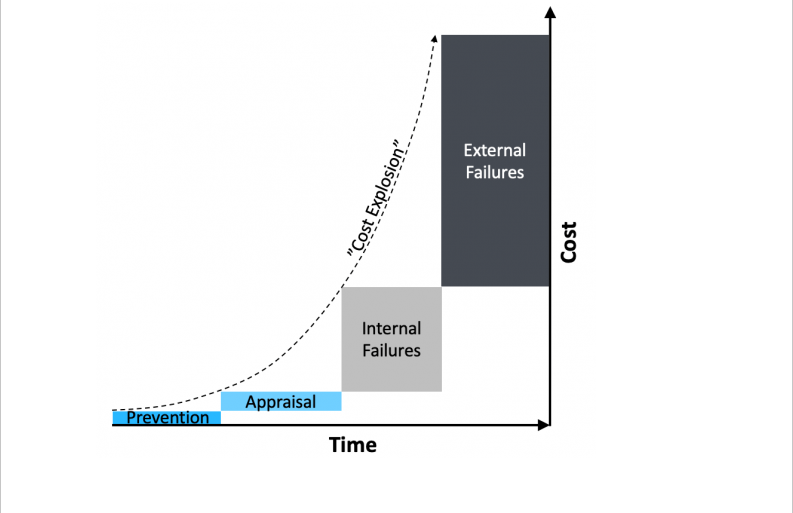
Reactive Quality Management
Problem: dealing with failures once they have occurred and costs already “exploded”
Costs balloon by a factor of 10x each time a problem escapes detection. Quality excellence therefore requires shifting focus upstream, allocating resources to controllable quality costs:
➤ Error-proofing (prevention cost)
➤ Failure detection (appraisal cost)
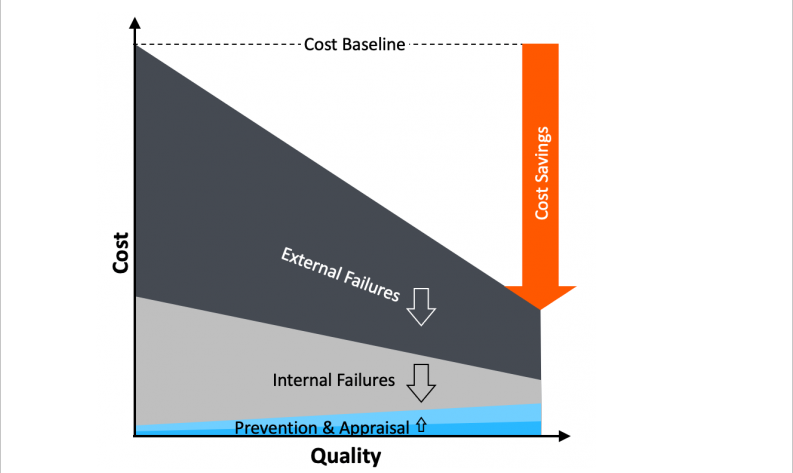
Quality Excellence Management
Solution: dealing with variability early to prevent costly failures downstream
Quality excellence means front-loading efforts, controlling critical-to-quality (CTQ) parameters upstream, so that major failures can be prevented downstream, significantly reducing resultant quality costs:
<span style=”color: #99cc00;”>➤ </span>Scrap and rework (internal failure cost)
<span style=”color: #99cc00;”>➤ </span>Returns and claims (external failure cost)
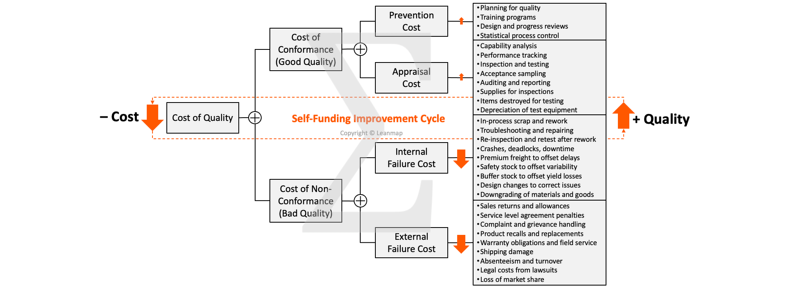
Analyze Quality Costs to Prioritize Improvement Actions
➤ Quality Costs are those associated with preventing, detecting, and remediating problems to meet the expectations of a customer, the good and bad quality costs:
➤ Cost of Conformance to ensure good quality:
➤ Prevention Cost involves all activities to minimize the potential for defects, including planning for quality, operator training, design and progress reviews, statistical process control, and supplier certification.
➤ Appraisal Cost involves all activities to detect problems early; it covers labor and overhead for capability analysis, performance tracking, inspection and testing, acceptance sampling, auditing and reporting, supplies required for inspections, items destroyed for testing, depreciation and maintenance of test equipment.
➤ Cost of Non-Conformance to correct bad quality:
➤ Internal Failure Cost involves all activities to detect a defective item that needs to be reworked and retested or scrapped if deemed unusable; it covers in-process scrap and rework, trouble-shooting and repairing, re-inspection and rest after rework, as well as crashes, deadlocks, breakdowns, downtime, premium freight to offset delays, safety stock to offset process variability and inflexibility, buffer stock to offset yield losses, design changes to correct product instabilities, downgrading of materials and goods because of imperfections.
➤ External Failure Costs involves all activities to deal with a defective item that reached the customer, including sales returns and allowances, service level agreement penalties, complain and grievance handling, product recalls and replacements, warranty obligations, field service, shipping damage, absenteeism and employee turnover, legal costs from lawsuits, loss of market share.
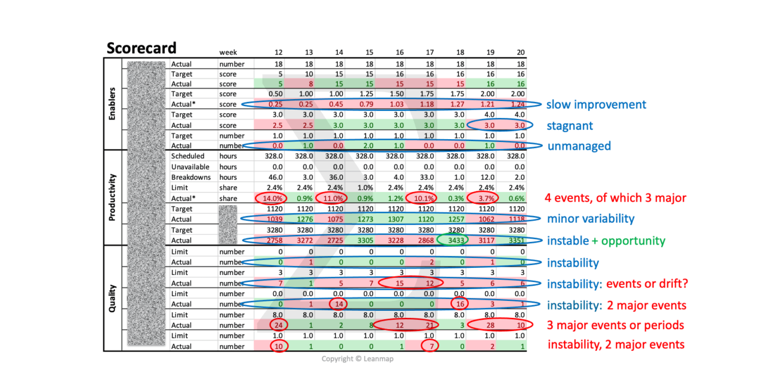
Quality Performance Review Based on Scorecard to Identify Abnormalities
The balanced scorecard is a strategic planning and management tool to communicate plans and targets, schedule resources, prioritize quality improvement actions, and monitor progress towards strategic quality targets. The scorecard is “balanced” when reviewing operational measures and performance enablers, in addition to financial measures. The framework gives an organization a way to “connect the dots” between quality planning, performance measurement, performance management, and impact verification based on key performance indicators (KPIs) to accomplish the quality mission, vision, and strategy. Below an example of an manufacturing scorecard that focuses on three areas of performance improvement:
➤ Enablers: skill score, workplace organization, problem-solving effectiveness
➤ Productivity: units per man-hours, units per machine-hour, output per week
➤ Quality: abnormalities, rejects, scrap, downtime, and customer complaints
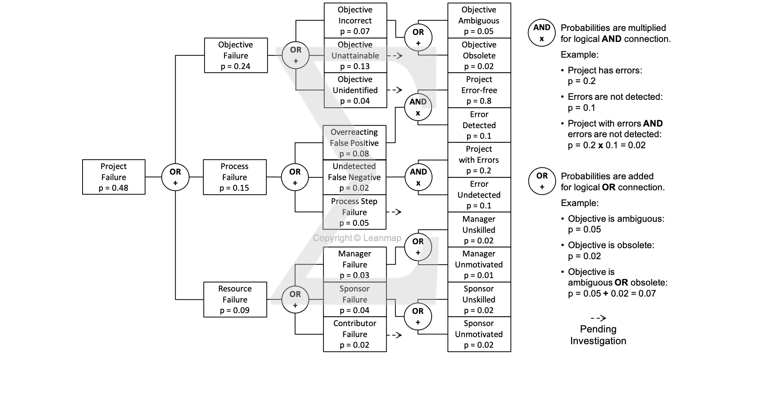
Project Failure Analysis to Identify Root Causes Using the Defect Tree
The logic tree is a key tool to analyze defects, uncover their causes, and develop quality improvement actions. The example below analyzes the causes for unsuccessful projects: 48% projects failed due to defects in objectives (24%), processes (15%), and resourcing (9%). The analysis shows that ambiguous objectives caused 5% project failures. By clarifying objectives, using the SMART framework, this cause can be effectively addressed in the quality improvement plan.
Quality Excellence in Health Care
Measuring and monitoring overall system performance is challenging and complex but critical to improve the quality of patient care. Doctors and hospitals are increasingly being held accountable to develop actions aimed at improving the quality of care, reducing costs, and achieving better patient-centered care. This following QI project case describes how to analyze a clinical care issue and steer quality improvement efforts to achieve better patient outcomes.
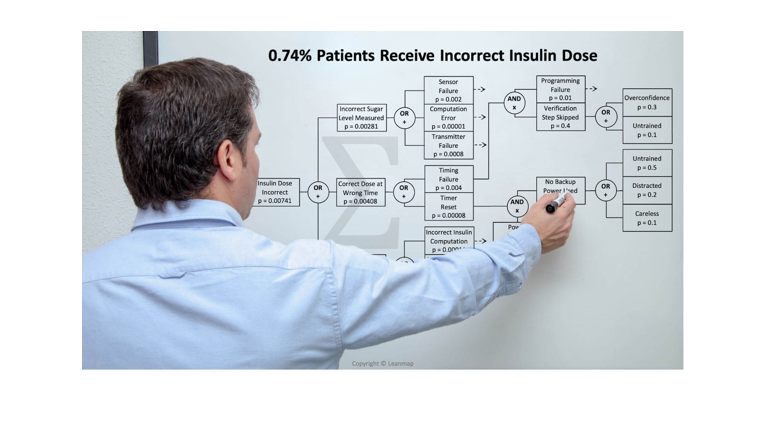
Incorrect Insulin Dose Administered to 0.74% Patients
The fault tree analysis shows three physical causes for administering the incorrect insulin dose: (a) incorrect sugar measurement from sensor failure, computation error, or transmitter failure, (b) correct dose administered at the wrong time because of timing failure or timer reset, and (c) delivery system failure because incorrect insulin computation, incorrect pump signal, or pump failure. When investigating the timing failure, we found it was caused by a combination of programming failure and verification step skipped. And skipping the verification was due to overconfidence or lack of training. And the timer reset was caused by a power outage and the failure to connect the backup power, which was caused by the health care worker being distracted, careless, or overconfident that a power outage will not happen. By analyzing the causes at the physical, human, and latent level, we help clinicians to develop effective countermeasures that improve patient safety and overall health care quality by 10x to 100x.
